PERIODIC SYSTEM
According to J. Jungius (1630) and R. Boyle (1661), chemical elements are 'those substances that cannot be broken down into anything simpler' In 1803, J. Dalton postulated his atom theory that each element was composed of tiny indivisible particles, the atom (from the Greek ·tomos, indivisible). The atoms are distinguished mainly by their weight, in which the ratios of the atom weights of various elements can be approximately described by integer proportions (e.g. 3:2). The result of this is that the elements are classified and numbered according to their increasing atom weight (see footnote 1) which is more or less comprehensible even to the listener who has not had a scientific education, without any claims to absolute scientific correctness.
Closer investigation revealed that there is a type of 'chemical bond' between the elements, i.e. that the chemical properties repeat themselves in a series of eight elements. In 1911, E. Rutherford discovered that an atom is made of a tiny nucleus surrounded by electron shells, which is responsible for the behaviour of an element. These electron shells have as many electrons as the atom has positive charged protons in its nucleus, so that the atom is electrically neutral. The number of the protons in the nucleus is decisive for the classification of an atom to a particular element. The electron shell has a shell-shaped structure in which each main shell is made up of subshells (orbital / orbital types s, p, d, f; this subshell is made up of further subshells (the s orbital has no subshells, the p orbital has 3 subshells, the px, py and pz orbitals; the d orbital has 5 subshells, the f orbital has 7 such subshells). Each one of these subshells has a maximum of 2 electrons which are distinguished by their spin (a property of the electrons which make them noticeable in a very magnetic field) (--> Pauli exclusion principle). In general, each electron of the electron shell is characterised by a series of four so-called quantum numbers (the principle quantum number and 3 orbital quantum numbers; for the orbital shape, the magnetic quantum number and the spin) with which a certain discrete energy is connected to the individual electrons. This energy corresponds to a characteristic spatial form of the electron orbits or rather, more specifically, the probable position of the electron.
1. CHRONOLOGICAL STRUCTURE
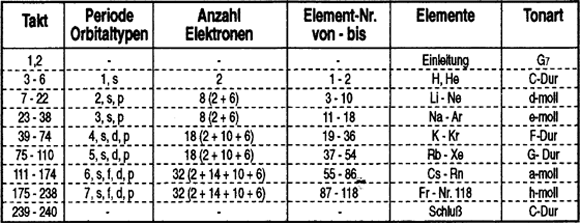
The composition follows the sequence of the elements according to their atomic numbers, starting with hydrogen (AN 1), through to helium (AN 2), lithium (AN 3) etc. right through to the final known element number 109 (current name: Meitnerium). Furthermore, for musical reasons, the periodic system is expanded to the ñ as yet undiscovered element 118 (--> surround of the seventh shell). A two-act theme or a musical phrase derived from such is allocated to each of the elements. The resulting 236 bars are supplemented by two additional preludes / final bars. Parallel to the transition of the periods, there is a change of tone which is orientated towards the triad stages of a C major scale. Table no. 1 summarises the chronological structure.
Table no. 1:
The periodic system of the elements as a chronological / harmonious structural principle of the Periodic System composition. The types of orbital are given for the appropriate period in the sequence in which they appear. All shells commence with the elements whose s orbitals are occupied (alkalis and alkaline earths). In the example of the fourth period, in the case of 10 elements, first of all the d orbitals will be occupied, and the shell is concluded with the occupation of 6 p orbitals.
2. MUSICAL INTERPRETATION OF THE TYPE OF ORBITAL ñ
CONFIGURATION OF THE THEME AND SHAPE OF THE ORBITAL
Decisive for the chemical behaviour of an element is the state of occupation and the geometry of the valence orbital i.e. of the external electrons. The shape of the orbitals are reminiscent of Chladni's figures and, like them, are distinguished by the number of their so-called nodal lines / levels. Two decisions were crucial to the development of the musical themes for the individual types of orbital: 1) reminiscent of Pauli's exclusion principle, there was to be no repetition of the tone within one octave
2) the nodal levels were to be realised by means of a specific sequence of scale figure and interval jump. The results are displayed in table no. 2
3. MUSICAL INTERPRETATION OF THE MAGNETIC QUANTUM NUMBER OF THE ELECTRON SPIN
Several so-called magnetic quantum numbers can be distinguished within one type of orbital ñ as shown in table no. 2. The magnetic quantum number has been interpreted as the changing of the theme in the fifth circle regarding the tone of each prevailing period, whereby a negative sign indicates a fifth pace downwards and a positive sign indicates a fifth pace upwards (e.g. 6th period (A minor) quantum number ñ2 --> theme in G minor). The electron spin is characterised by the theme (for spin up) / by the return of the theme, i.e. the theme backwards (for spin down).
4. INSTRUMENTATION AND MATERIAL PROPERTIES
If you follow the instrumentation key in table 2, there ensues an instrumentation orientated towards the element groups: change of orbital within a period and change of period are outlined by the timpanist.
| s-orbitals |
p-orbitals
|
d-orbitals
|
f-orbitals |
| alkaline / chalcogens |
non - metals |
metals |
actinides / lanthanides |
| strings |
wood winds |
brass |
piano |
For elements with p and d orbitals, the instrumentation is further differentiated:
Table 2:
| p - orbitals |
volatile non-metals
solid non-metals
semi-metals
elements - metallic character |
flute
claninet
oboe, horn
bassoon |
| d - orbitals |
soft metals
hard brittle metals |
horn
trumptet |
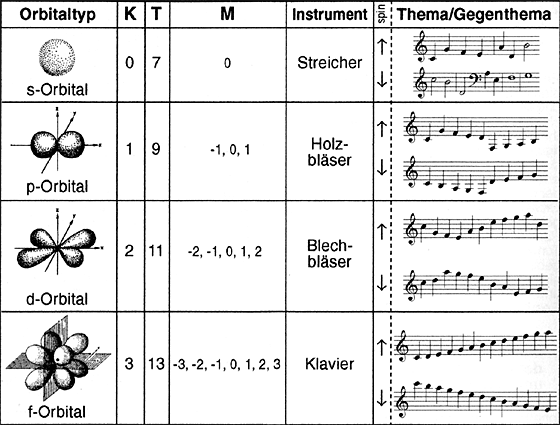
Type of orbital, geometry, corresponding themes and instrumentation in the Periodic System composition. Column K: amount of nodal levels for the individual types of orbital. Original meaning: number of jumps (interval > long seconds) in the theme with the exception of the s orbitals. For the brass section, there is a surplus jump in the theme (reference to the differing geometry of the dz2 orbital). A simple scale without jumps was chosen for the f orbitals (characteristic distinction from the s, p and d orbitals). T: number of tones in the theme, M: magnetic quantum numbers (in this composition, the quantum numbers have been interpreted as the number of stages in the fifth circle): Theme / counter theme both start from C, indicated rhythmically indifferently for p, d and f orbitals. Octave transpositions on jumps are allowed due to the position of the performing instrument.
5. HANDLING OF THE INNER ORBITS
Only the newly-entering electron with its orbital has been considered in the musical version of the elements so far. However, these newly-entering electrons are also present in the following elements, forming the so-called inner orbits. In the musical interpretation, the theme repeats itself once more within the period; however its rhythm is changed and sometimes segmented. In each following period, the theme is once again repeated in a changed, transposed and segmented form and adapted to ìfit intoî the overall musical sequence. The musical priority sets the prevailing musical theme. The segmented and transposed material which represents the orbitals and the elements of the preceding periods thus forms a musical, harmonious background for each prevailing melodic occurrence.
j.s
|